
2.1 GA structures on black shale bedding surface. Scale bar 1 cm. (El Albani et al. 2010)
The latest edition of Nature has a paper claiming 2.1 billion year (Ga) old multicellular colonial organisms from Gabon. This is a remarkable claim, since the oldest definitive large multicellular organisms so far found in the fossil record are from less than 600 million years ago (It is still not certain if the 2 Ga Grypania spiralis is a eukaryote or large bacterial colony). So remarkable is the claim in fact, that it made the front cover of Nature.
I think they are wrong.
Lets be more specific here. There are two claims - that the structures are 2.1 Ga old, and that they represent large multicellular colonial organisms.
First the date. That appears well constrained. Chris Rowan has good coverage of the dating at Highly Allochthonous, so I have no argument against the age.
I don’t think they are multicellular colonial organisms. I think they are pyritised remains of microbial mats, or structures associated with microbial mats.
There’s been ample coverage of the paper, but I want to concentrate on the reasons why the authors think that the structures represent organised colonial organisms.
The structures are found in unmetamorphosed black shales. Over 250 specimens have been recovered. They are pyritised and embedded within the sediment. They range from 7-120mm in length, 5-70 mm in width, and 1-10 mm thick. They can occur in densities of up to 40 specimens per square metre, at random orientations, but all are horizontal to bedding. All are wrinkled to some degree, with some having large central pyrite nodules. Some show significant folding. All show radial cracks. The authors used micro-computed tomography (Micro-CT) to produce three-dimensional images of the structures. They also undertook carbon and sulphur isotope analyses of the host rock and the structures.
On the following lines of evidence the authors decided that the structures represented multicellular colonial organisms:
- The structures shown under Mirco-CT are not the same as produced by microbial mats.
- They are unaware of any inorganic process that could produce the folding patters seen in the structures.
- The folding suggests an originally cohesive flexible sheet.
- They are unaware of any inorganic process that could produce the radial fabric seen in the structures.
- The radial fabric represents peripheral accretion of flexible organic matter.
- The 12C/13C carbon isotope ratio in the host sediment is different to that in the structures suggesting the structures represent distinct organisms.
- Steranes have been found in the shales, a compound associated with eukaryotes.
- A lack of support for any other inorganic origin.
I think pyritisation of microbial mats is a better explanation. Here’s why.
I’d like to take 1, 2, and 3, together because they all relate to the physical properties of microbial mats.
First let's take a look at what we're talking about.
y.jpeg) Micro-CT-based reconstructions and virtual sections of four specimens from the FB2 member of the Francevillian Group. First Column - original. Second column - volume rendering in semi-transparency. Column 3 - Transverse two-dimensional specimen. Column 4 - longitudinal section running close to estimated centre of specimen. Scale 5 mm. (El Albani et al. 2010)
Micro-CT-based reconstructions and virtual sections of four specimens from the FB2 member of the Francevillian Group. First Column - original. Second column - volume rendering in semi-transparency. Column 3 - Transverse two-dimensional specimen. Column 4 - longitudinal section running close to estimated centre of specimen. Scale 5 mm. (El Albani et al. 2010)The structures are pretty much all pyrite. The absence of pyrite is marked by the radial cracks. The bright central area in rows 3 and 4 represent a central pyrite nodule. This is not present in all specimens. This is important, as the large thickness values for specimens are all associated with this central nodule. Away from the nodule, or when a nodule is not present (e.g. row 2 above), the structure is a thin film of pyrite around 1-2 mm thick.
This is really important. All the folding is just that, folding. The apparent holes (black areas) in the images above do not represent complex structure within the specimen. The image is a slice through the specimen at a certain level. Where the pyrite film is folded below the level being imaged (downward folds), or completely above it (upward fold), you get a black space. The images appear to show complex structure, but it is simply a line drawn across an undulating surface. There is no internal structure here other than the radial fabric.
Further, and critically, the authors have concluded that the thin pyrite film and the central nodule represent different periods of pyrite formation. This is supported by sulphur isotope data, and I am happy to accept their conclusion.
This means that the large nodules have little to do with the original structure of the specimens, and, in fact they appear to occupy a central cavity in the specimen when they are present. An example of an actual nodule is below.
 Section through a specimen showing central pyrite nodule surrounded on both sides by sheet material. Scale bar 1 mm. (El Albani et al. 2010)
Section through a specimen showing central pyrite nodule surrounded on both sides by sheet material. Scale bar 1 mm. (El Albani et al. 2010)In this specimen you can see that the nodule and the thin film on either side of it is almost all pyrite. The gaps in the center of the nodule are unexplained, but probably host sediment. Ignore the pretty colours. that refers to sulphur isotope analysis which I'm not questioning.
So, if we ignore the central nodules for the present, we are left with an original structure that is a thin film approx. 1-2 mm thick which has been thrown into folds in some specimens (e.g. row 2 above) but not in others (e.g. row 1 above).
The authors claim that the folding and the radial cracks cannot be accounted for by inorganic processes. I think they can.

Look! over there on the right! Is a 2.1 Ga colonial organism with a radial fabric? Is it a Proterozoic jellyfish? No, it's . . well, I'll explain later. But compare this example with rows 2, 3 and especially 4 of the Micro-CT images above. Notice that it has a central zone which is distinct from the periphery. Notice there is a faint dark band separating the central core from the periphery. Notice also the distinctive radial 'cracks' that spread out from the core Central mass? Check, radial cracks? Check. This would appear to be a good match to the 2.1 Ga structures.
But if you are not convinced, here's another variety of the same structure. this time we'll compare it directly with row 2 above as this has the best fold structure presented.



Can you see the fold pattern at the centre of the lower photos? It's quite similar to the fold pattern in the Micro-CT image. These images are of water escape structures in Ediacaran sandstones. The difference in colour you can see is due to the sandstone being deposited wet and dirty - i.e. waterlogged and with a significant amount of silt and clay (the red stuff). Escaping water carries the red silts and clays away with it, leaving the white sand behind.
Three things to note. They can produce radial structure, they can produce fold-like structures, and they are pretty similar in size even though they are from different sandstones.
Now, for the record, I don't think that the radial structures in the 2.1 Ga structures are water escape features (I've another idea for them), I'm just showing that such structures can be formed inorganically. Although I am suggesting that water escape could be an explanation for the folding we see in the row 2 specimen.
The authors discount water escape as an explanation because the underlying sediment shows no disruption such as that shown in my Ediacaran example viewed from the side. Plus, shales are not known for their permeability (ability to allow water to flow through it). But we are dealing here with microbial mats. Mats can act as a barrier to water or gas flowing below them. In such instances, rapid loading by waning storm sediments could cause water or gas trapped under the microbial mats to tear through the mat, creating some neat folding patterns and not disturb the underlying sediment too much.
Actually I'd better show you some mats so that you know what I'm talking about. Here's one.
 A juicy, yummy microbial mat, full of cyanobacterial goodness from Yellowstone Park. Photo Carnegie Institution
A juicy, yummy microbial mat, full of cyanobacterial goodness from Yellowstone Park. Photo Carnegie Institution(And yes I do consider Yellowstone to be a good analogue for the 2.1 Ga Proterozoic, because while the hydrothermal pools are aggressive to microbial mat grazers, so was the 2.1 Ga Proterozoic, because, well, there weren't any.)
Do you think the microbial mat above looks like what the authors have imaged? I do.
And just so we're clear on how flexible microbial mats can be, here's another photo.
 Photo from Cuadrado and Pazini (2007)
Photo from Cuadrado and Pazini (2007)Here are some more Microbial mats.
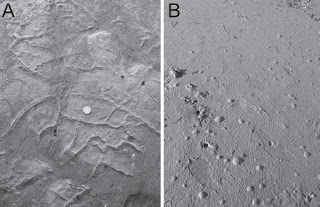 Photo from Cuadrado and Pazini (2007)
Photo from Cuadrado and Pazini (2007)Photo A shows microbial mats draped (folded) over cracks. Photo B shows gas bubbles trapped under a microbial mat. What? you thought I just made that gas stuff up?!
But this is the clincher for me.
 Microbial mats at West Chaplin Lake (Bowman and Sachs 2008)
Microbial mats at West Chaplin Lake (Bowman and Sachs 2008)There are a number of things to notice here. Firstly the mats are not one continuous sheet, but here are two discrete mats about the same dimensions as the 2.1 Ga structures. Also note the lower mat has been torn to produce a clean, sharp, high angled edge. This shows that these things can be torn without losing structure, so simple folding should be no problem. Speaking of folding, check out the left margin of the upper mat. See the fold? Remind you of anything? This maybe?

So microbial mats can fold and wrinkle without tearing. I agree with the authors that the structures represent originally cohesive flexible sheets, but microbial mats not colonial multicellular organisms.
There are other methods to fold or wrinkle microbial mats. Storms could rip up mats or partially dislodge them for example, and the authors interpret the environment as deltaic, influenced by storms. Elephant skin textures on bedding plains have been interpreted as being caused by wrinkling and folding microbial mats.
I hope this has shown that the structures seen in the 2.1 Ga specimens, the folding and wrinkling, can be produced by inorganic means on biological sheets without invoking a new class of organism.
OK, on to numbers 4 and 5, the radial fabric.
This is one of the main arguments for the structures being a colonial multicellular colonial organism and it certainly is striking. but, as I showed above, radial fabrics can be caused by other things. inorganic things.
This irregular radial fabric, along with the the fact that it is commonly deflected to meet the edge of the structures is taken as evidence that the structures grew by peripheral accretion of flexible organic matter.
However, there are a few specimens where this radial fabric appears to go right through the structure.
 Scale bar 1 cm
Scale bar 1 cmIn one example, the fabric appear to actually cut through the central nodule
 Scale bars 5 mm
Scale bars 5 mm
How could a growth-related fabric cut through a nodule that wasn't there until late in the diagenetic process?
I think it's down to water again.
Lets assume that the mats/colonial organisms have gone through initial diagenesis and have been converted to pyrite. As diagenesis proceeds and load on the sediment column increase, the mudstones start to compact. Muds can contain 60% water which means that they have a long way to compact. Problem. Pyrite sheets can't compact.
I think that as compaction progressed and the pressure increased, the thin outer margin split in a regular pattern. As the process continued, the cracks spread further into the body of the specimens and sediment would be forced into the cracks. As we have an example of crack through the central nodule, it may be that this cracking occurred late in the diagenetic process, when compaction was reaching it greatest extent.
So rather than be evidence of peripheral growth, the fabric may be diagenetic.
Number 6 and 7 are linked, 6 is the carbon isotope ratio, and 7 the presence of steranes.
The authors found a difference in the organic carbon isotopic composition between the host rock and the structures. This, they say, shows that the structures were distinct organisms.
Without going into too much detail, carbon is present in two main isotopes 12C and 13C with one extra neutron in the nucleus. Organisms preferentially take up 12C in different amounts depending on their metabolic pathways, causing a shift in the 12C/13C ratio. Measuring this difference or delta 13C can help identify the origin of the organic mater.
The host rock has a delta 13C value of -27 while the structures had a value of -32. Now -27 is roughly an average value for eukaryotes, and so would be expected to represent the fallout from the water column into the sediment of dead eukaryote plankton. A value of -32 is more like bacterial signature. This would tend to support my suggestion that these structures are pyritised microbial mat fragments.
Number 7 is the observation that steranes have been found in the shales (note the shales not the structures). Steranes are compounds associated with eukaryotes. But if the explanation for the delta 13C values is correct, the steranes would also be produced from the eukaryote plankton and can't be used to support the suggestion that the structures are eukaryotes.
Finally number 8, a lack of support for any other inorganic origin.
I think I've put up enough suggestions for inorganic input into the formation of these structures. but here is a rough explanation:
- Microbial mats grow on delta front muds - probably as individual round patches, possibly connected by thin connections to other mats.
- Storm activity rips up or distorts the mats into folds and waning storm currents deposit a layer of sediment over them.
- Pore fluids equilibriate and the mats are surrounded by sulphide dominated pore fluids.
- Pyrite replaces the mats.
- As the basin fills and the sediment column increases, the muds compact. The distortion and pressure casuse the pyrite structures to deform slightly and crack around the margins.
- As pressure and compaction increase the cracks propagate and second pyrite phase occurs in the central area of some structures. It may be that the cracks allow pore fluid and organic-rich sediment into the centre of the structure allowing the second pyrite phase to occur.
I'd like to commend the authors for taking a multidisciplined approach to this work. It's an approach which I believe should be done more often, and will be more widespread in the future.
I'd also like to commend them for their use of new techniques and I hope they will continue to use them to explore more of the geological record.
Sorry that this post has been a bit of a smorgasbord, but my aim is to present a number of possible scenarios for the formation of these structures that do not require that they be large, multicellular, colonial organisms. There are other, unfortunately more mundane, explanations.
UPDATE 1
UPDATE 2
Albani, A., Bengtson, S., Canfield, D., Bekker, A., Macchiarelli, R., Mazurier, A., Hammarlund, E., Boulvais, P., Dupuy, J., Fontaine, C., Fürsich, F., Gauthier-Lafaye, F., Janvier, P., Javaux, E., Ossa, F., Pierson-Wickmann, A., Riboulleau, A., Sardini, P., Vachard, D., Whitehouse, M., & Meunier, A. (2010). Large colonial organisms with coordinated growth in oxygenated environments 2.1 Gyr ago Nature, 466 (7302), 100-104 DOI: 10.1038/nature09166
Diana G. Cuadrado and Natalia V. Pizani. (2007) Identification of microbially induced sedimentary structures over a tidal flat. Latin American Journal of Sedimentology and Basin Analysis. v.14 n.2 La Plata ago./dic. 2007
Jeff S Bowman and Julian P Sachs (2008) Chemical and physical properties of some saline lakes in Alberta and Saskatchewan. Saline Systems, 4:3 DOI: 10.1186/1746-1448-4-3

 The specimen at right (Fig. A) is a Meraspid 1, the smallest meraspid so far found at Emu Bay.
The specimen at right (Fig. A) is a Meraspid 1, the smallest meraspid so far found at Emu Bay. As the meraspid grows, segments are added from the front of the tail. We can see this in meraspid 7 at right (Fig. B, scale bar 1 mm). This has 7 segments, but the two original segments with the spines can be seen at the front of the thorax, with the non-spiney segments, which have been added later, positioned behind these two. If the thorasic segments were added from the back of the head, then the first two segments with spines would be at the back of the thorax next to the tail. The large spines on the head are also still very visible.
As the meraspid grows, segments are added from the front of the tail. We can see this in meraspid 7 at right (Fig. B, scale bar 1 mm). This has 7 segments, but the two original segments with the spines can be seen at the front of the thorax, with the non-spiney segments, which have been added later, positioned behind these two. If the thorasic segments were added from the back of the head, then the first two segments with spines would be at the back of the thorax next to the tail. The large spines on the head are also still very visible. In Meraspid 10 at right (Fig. C, scale bar 1 mm) things are starting to change. The first thing to notice is that the large spine on segment 1 is almost gone. There is a much reduced spine there. Also the spines have become more curved. They are still long though - still the length of the meraspid. The head is very well preserved and provides a wealth of information on the way the various elements of the head change with growth. The rounded shape and the spines are very distinctive. Nothing else in the fauna is like that. So, even though it's only a few millimetres across, it immediately caught my eye.
In Meraspid 10 at right (Fig. C, scale bar 1 mm) things are starting to change. The first thing to notice is that the large spine on segment 1 is almost gone. There is a much reduced spine there. Also the spines have become more curved. They are still long though - still the length of the meraspid. The head is very well preserved and provides a wealth of information on the way the various elements of the head change with growth. The rounded shape and the spines are very distinctive. Nothing else in the fauna is like that. So, even though it's only a few millimetres across, it immediately caught my eye. Finally we have meraspid 12 (Fig. D, scale bar 1 mm). We are almost at the Holaspid stage or mature stage, of 13 segments, and the meraspid is now similar to the final adult shape. Note that the second thorasic spines are much reduced, and the spines on the head are also beginning to reduce in length. The thorasic spines will completely disappear in the adult, and the spines on the head will be reduced, but still present.
Finally we have meraspid 12 (Fig. D, scale bar 1 mm). We are almost at the Holaspid stage or mature stage, of 13 segments, and the meraspid is now similar to the final adult shape. Note that the second thorasic spines are much reduced, and the spines on the head are also beginning to reduce in length. The thorasic spines will completely disappear in the adult, and the spines on the head will be reduced, but still present.

 This is a plot of the length of the longest part of the head (cranidium) against the width from eye to eye, and the longest part of the head against the longest part of the glabella - the central bulb area of the head.
This is a plot of the length of the longest part of the head (cranidium) against the width from eye to eye, and the longest part of the head against the longest part of the glabella - the central bulb area of the head.










y.jpeg)












